COVID-19 Response
We are proud to support the COVID-19 response in China and around the world.

We are proud to support the COVID-19 response in China and around the world.

Our COVID-19 vaccines are protein-based subunit vaccines. Like all vaccines, they contain molecules from the target pathogen that trigger an immune response and create pathogen-specific “memories.” With these memories, the body is trained to protect itself against the pathogen and prevent the disease that it causes.
Subunit vaccines contain purified pieces of the target pathogen that have been specifically selected for their ability to stimulate immune cells. These fragments are incapable of causing disease. Our protein-based subunit COVID-19 vaccines are based on the spike (S) protein, which is located on the viral envelope of SARS-CoV-2. The S protein is responsible for binding to and entering the cell, making it the primary target antigen for vaccine development. Protein-based subunit vaccines can trigger the immune system to destroy the pathogen and cells that have been infected by it. Here's how: cells infected by SARS-CoV-2 will process the virus and put its S-protein fragments on their surfaces, which activates a type of immune cells called killer T cells. Killer T cells recognize SARS-CoV-2-infected cells and destroy them before they have a chance to produce new viruses.
Protein-based subunit vaccines may also create a group of immune cells called memory B cells and memory T cells. When the body encounters the SARS-CoV-2 virus, it will remember to generate new antibodies and mount a T cell response to counterattack the virus. This type of memory allows for a vaccine to generate longer-lasting protection from disease.
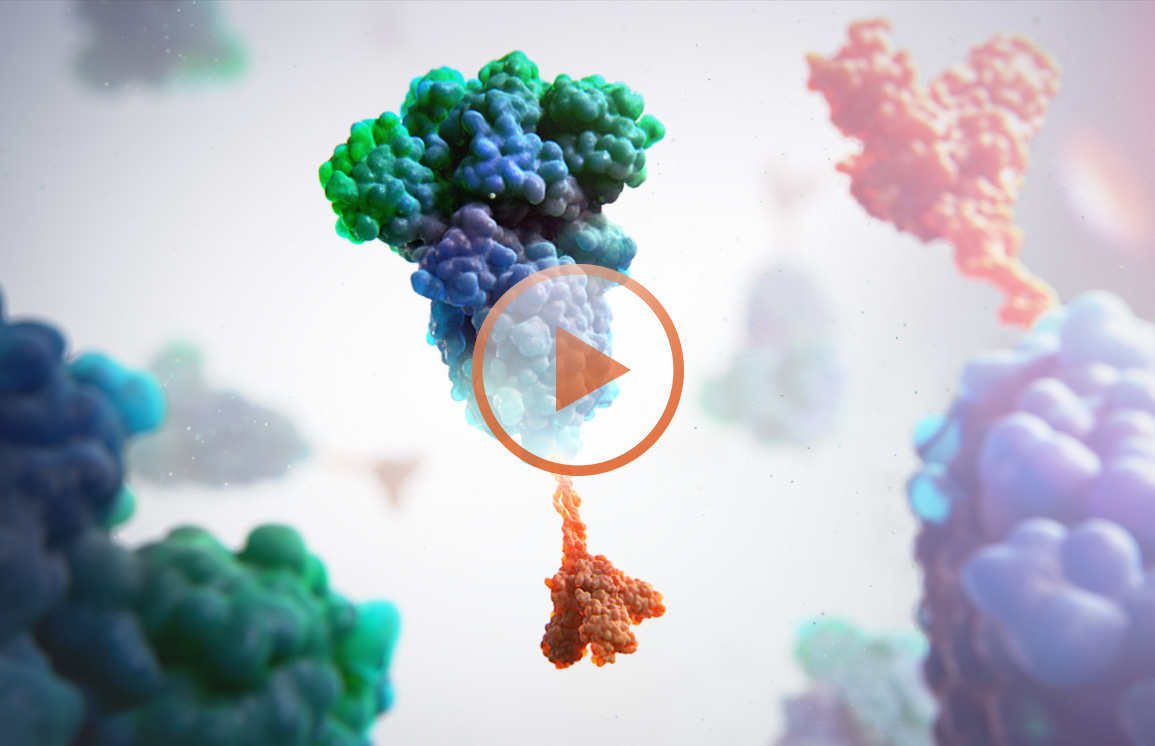
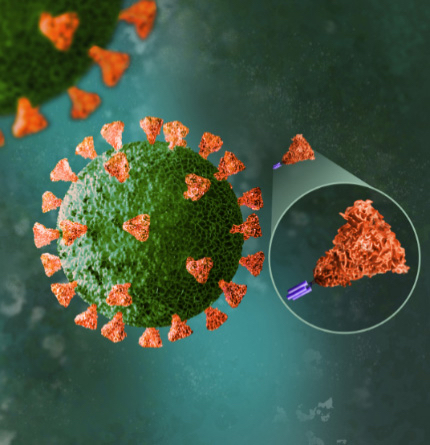
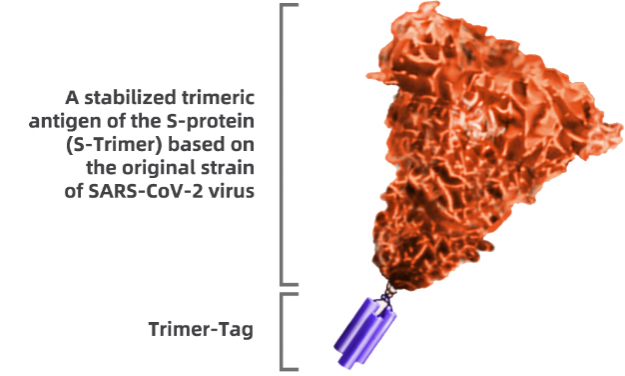
SCB-2019 (CpG 1018/Alum) consists of two key components: SARS-CoV-2 antigen and adjuvant. Leveraging the Trimer-Tag technology platform, we developed the SCB-2019 antigen based on the S protein in the original strain of SARS-CoV-2. Our antigen is a recombinant SARS-CoV-2 fusion protein that remains stable in its original form, allowing it to elicit protective immunity against the SARS-CoV-2 virus.
To further help the immune system respond to our antigen, we add two adjuvants: CpG 1018, an advanced adjuvant developed by Dynavax Technologies, and aluminum hydroxide (alum). Both adjuvants are proven technologies currently used in commercialized vaccines that U.S. and European Union authorities have approved. They are produced at scale under conditions that meet international standards for quality and excellence and have significant safety databases in clinical and post-marketing studies.
Our publications provide additional information on our COVID-19 vaccine development.
LEARN MORE>>